Brand Name: Furocef 750
Manufacture: Renata Limited
PRESENTATION
Furocef ® 125mg tablets : Each film coated tablet contains 125 mg of cefuroxime (as
cefuroxime axetil USP). Furocef ® 250mg tablets : Each film coated tablet contains
250mg of cefuroxime (as cefuroxime axetil USP). Furocef ®¨ 500mg tablets : Each film
coated tablet contains 500mg of cefuroxime (as cefuroxime axetil USP). Furocef ®
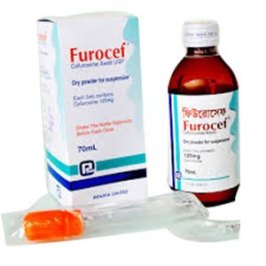
powder for oral suspension : Provided as dry, white to pale yellow powder. When
reconstituted as directed, each 5 ml of suspension provides the equivalent of 125 mg of
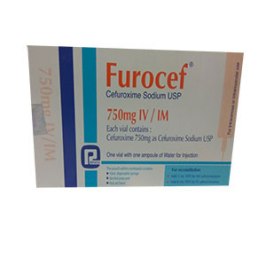
cefuroxime (as cefuroxime axetil USP), Furocef ® 250mg IV/IM Injection : Each vial
contains 250mg of cefuroxime (as cefuroxime sodium USP). Furocef ® 750mg IV/IM
Injection : Each vial contains 750mg of cefuroxime (as cefuroxime sodium USP).
Furocef ® 1gm IV/IM : Each vial contains 1g of cefuroxime (as cefuroxime sodium USP).
Furocef ® 1.5gm IV : Each vial contains 1.5g of cefuroxime (as cefuroxime sodium USP)
SIDE EFFECTS
General : Anaphylaxis, angioedema, pruritus, rash, serum sickness-like reaction and
urticaria. Gastrointestinal: Pseudomembranous colitis. Hematological: Haemolytic
anaemia, leukopenia, pancytopenia, thrombocytopenia and increased prothrombin time.
Hepatic: Hepatic impairment including hepatitis and cholestasis, jaundice. Neurological:
Seizure. Skin: Erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal
necrolysis. Urological: Renal dysfunction. Cephalosporin-class adverese reactions:
Toxic nephropathy, aplastic anaemia, haemorrhage, increased BUN, increased
creatinine, false-positive test for urinary glucose, increased alkaline phosphatase,
neutropenia, elevated bilirubin and agranulocytosis.










Reviews
There are no reviews yet.